PLS HELP URGENT
Electron dot diagrams
Use your periodic table to write the electron dot diagrams for the following atoms.
1. Calcium (Ca)
2. Polonium (Po)
3. Moscovium (Mc)
4. Boron (B)
5. Fluorine (F)
Answers
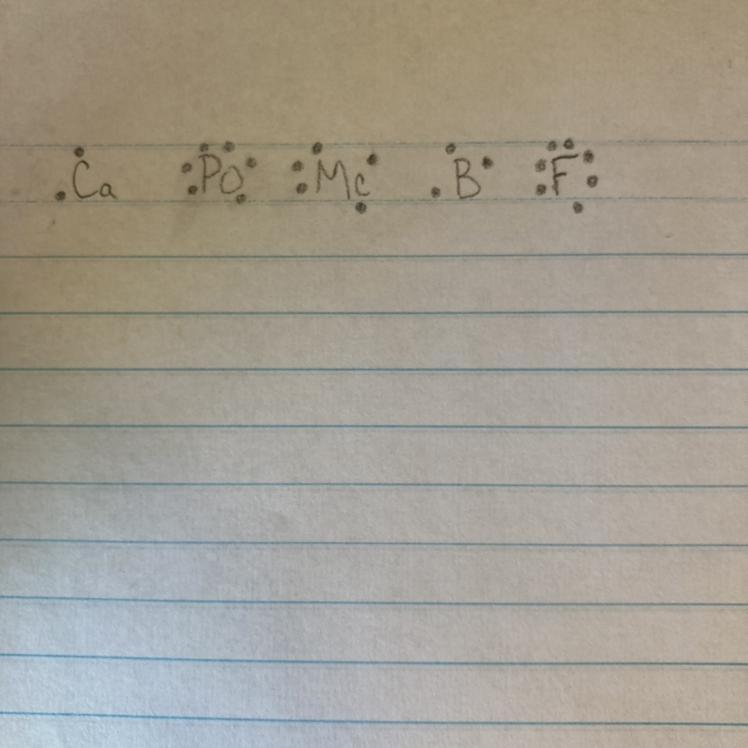
Related Questions
How many total atoms in 5 mg(NO3)2
Answers
Answer:
5 Mg (magnesium), 2 N (nitrogen), and 6 O (oxygen) -- total of 13 atoms
There are 45 as 1 mg(NO3)2 = 9 atoms.
How many molecules are in 1 mole of molecules?
Answers
Answer:
6.022x10^23
Explanation:
How many ions are in the following compounds?
a. (NH4)3PO4 =
b. NaCl =
Answers
Answer:
A- In (NH4)3PO4 there are four ions in total as three ions of NH₄ and is one ion of PO₄
Pleaseeee help pretty please
Answers
Answer:
A object made from iron .
Explanation:
बा
If OPPOSITE poles (N and S) of
a magnet are placed near
each other, what happens?
Type answer here
Answers
Answer:
They attract each other
Explanation:
A magnet has two poles;
North pole and south pole
When two opposite poles are placed close to each other, they attract each other. The short rule is opposite opes attract.
When two like poles are placed close to each other, they repel each other. The short rule is like poles repel.
what happens when bromine water is added to ethane and ethene
Answers
Answer:
Bromine water reacts with ethane as it is able to break open the double bond, forming 1,2-dibromoethane. This double bond is not present in ethane, so bromine water does not react with it. This reaction is known as electrophilic addition.
Explanation:
Ethene will readily react with bromine, so the colour of the bromine water changes from red-brown to colourless. Bromine atoms will add across the double bond in ethene to produce just one product; 1,2-dibromoethane.
How many water molecules are in 5.2 moles of H20?
Answers
Answer: There are therefore 6.02 × 1023 water molecules in a mole of water molecules.
Explanation: Hope this helps
What temperature does neon condensate?
Answers
Answer and Explanation:
I believe −246.048 C or −411 F
what are the elements found in The human body?
Answers
The elements found in the human body would be the following:
1. hydrogen,
2. oxygen,
3. carbon
4. nitrogen
Explanation:They are found throughout your body, mostly as water but also as components of biomolecules such as proteins, fats, DNA, and carbohydrates.
Note to you:
Hope this helped and correct me if I am wrong, I did some outside research. Have a good one!
I NEED HELP PLZZ THX <3
Question 16
A hockey puck is hit with a large force. The puck moves at a high speed as it slides over the ice.
What happens according to Newton’s first law of motion?
A) The puck does not lose any speed because ice is a very slippery surface.
B) The puck gradually slows down because mass makes objects slow down.
C) The puck gradually slows down because friction would be acting on it.
D) The puck does not lose any speed because once an object starts moving, the force of friction would no longer apply to it.
Answers
Answer:
C) The puck gradually slows down because friction would be acting on it.
Explanation:
It would be C because the puck would be scraping the ice and when it's scraping it slowly losses its speed because the ground is pushing it lightly
Hoped that helped :/
DIMENTIONAL ANALYSIS!
‼️ASAP!!! BRAINLIEST!!‼️
PLS HELP!!! SHOW ALL WORK + STEPS!! Thx!
Answers
Answer:
1037.56 mph
Explanation:
We are told the distance around the earth which is the circumference is 40075 km.
Converting to miles gives 24901.451 miles
Formula for speed = distance/time
Time for one rotation is 24 hours.
Thus, speed = 24901.451/24 = 1037.56 mph
Nitrogen gas reacts with hydrogen gas to produce ammonia according to the following equation.
N2(g)+3H2(g) → 2NH3(g)
What is the mole of ammonia produced from 7.23 x 10−4 moles of the hydrogen reactant, assuming there is sufficient nitrogen to react?
7.23 x 10−47.23 x 10−4
8.42 x 10−38.42 x 10−3
4.82 x 10−44.82 x 10−4
4.83 x 10−3
Answers
Answer:
4.82 x 10^-4
Explanation:
7.23 x 10^-4 mol H2 l 2 mol NH3
------------------------------------------------------- =
l 3 mol H2
(7.23 x 10^-4) x 2 / 3 =
4.82 x 10^-4
Hope this helped! ;)
The mole of ammonia produced from 7.23 x 10⁻⁴ moles of the hydrogen reactant when there is sufficient nitrogen to react is 4.82×10⁻⁴.
What is moles?Moles is a unit which is used to estimate the amount of any substance and it is represented as:
n = W/M , where
W = given mass
M = molar mass
Given chemical reaction is:
N₂(g) + 3H₂(g) → 2NH₃(g)
In the question it is given that sufficient moles of N₂ is present, so the formation of product depends on the moles of H₂. From the stoichiometry of the reaction, it is clear that:
3 moles of H₂ = produce 2 moles of NH₃
7.23×10⁻⁴ moles of H₂ = produce 2/3 × 7.23×10⁻⁴=4.82×10⁻⁴ moles of NH₃
Hence, option (C) is correct i.e. 4.82×10⁻⁴.
To know more about moles, visit the below link:
https://brainly.com/question/1464305
Which is the Net Ionic Equation for:
(remember, the Net Ionic Eq. shows ALL of the PARTICIPANTS)
2FeCl3(aq) + 3Mg(s) ® 3MgCl2(aq) + 2Fe(s)
2Fe3+ (aq) + Cl3- (aq) + 3Mg(s) ® 3Mg3+ (aq) + Cl2- (aq) + 2Fe(s)
2Fe3+ (aq) + 6Cl- (aq) + 3Mg(s) ® 3Mg2+ (aq) + 6Cl- (aq) + 2Fe(s)
Answers
Answer: [tex]2Fe^{3+}(aq)+3Mg(s)\rightarrow 3Mg^{2+}(aq)+2Fe(s)[/tex]
Explanation:
Complete ionic equation : In complete ionic equation, all the substance that are strong electrolyte and are present in an aqueous state and represented in the form of ions.
Net ionic equation : In the net ionic equations, we do not not include the spectator ions in the equations.
Spectator ions : The ions present on reactant and product side which do not participate in a reactions. The same ions present on both the sides.
The complete balanced ionic equation will be:
[tex]2Fe^{3+}(aq)+6Cl^-(aq)+3Mg(s)\rightarrow 3Mg^{2+}(aq)+6Cl^-(aq)+2Fe(s)[/tex]
In this equation, [tex]Cl^-[/tex] are the spectator ions.
By removing the spectator ions from the balanced ionic equation, we get the net ionic equation.
The net ionic equation will be:
[tex]2Fe^{3+}(aq)+3Mg(s)\rightarrow 3Mg^{2+}(aq)+2Fe(s)[/tex]
Identify 5 factors of weather
Answers
Answer:
"The five factors are: the latitude zone, the elevation of the land, topography (landforms), ocean currents and winds."
Explanation:
Answer:
The five factors that determine the weather of any land area are: the amount of solar energy received because of latitude; the area's elevation or proximity to mountains; nearness to large bodies of water and relative temperatures of land and water; the number of such storm systems as cyclones, hurricanes, and thunderstorms resulting from air-mass differences; and the distribution of air pressure over the land and nearest oceans, which produces varying wind and air mass patterns.
Explanation:
What causes an object to have a positive charge?
Answers
Hey. Can someone, please help me with the following questions:
Satellites
Satellites ___________the earth.
You can see _____________ _______________ without a telescope.
An example of a man made satellite is the _____________ _____________ _____________.
The only natural satellite of the earth is the __________.
Choose from: moon, orbit, natural satellites, international space station
Planets
There are ________ planets you can see with the __________ __________.
The five planets are, _________, ___________, ___________, ___________, ___________.
The planets are part of the ___________ ___________.
Light from Saturn takes around ___________ hours to reach earth.
Choose from: 1,5, naked eye, solar system, Mercury, Venus, Mars, Jupiter, Saturn
Comets and Meteors
A _________ is a spectacular sight in the _________ sky.
They are _______ to see and could be described as giant ___________ that orbit the _______.
__________ are bits of dust and _________ that _______ up as they move through the earth’s ____________. A meteor that makes it to earth is called a ______________.
Galaxies
1. What are most of the dots of light that we see in the sky?
2. What is the name of our home galaxy?
3. What is the name of our nearest star?
4. How long does it take the light from the sun to reach the earth?
5. Can you say what some of the other dots of light in the night sky might be?
6. How many stars might there be in one galaxy?
7. What is the name of our nearest neighbor galaxy?
8. State how long it takes for the light from our neighbor galaxy to reach the earth
Answers
5, naked eye, Mercy, Venus, Mars, Jupiter, Saturn, solar system, 1
I would need to see the choices for this section
1.Stars
2. The Milky Way
3. The sun
4.8 minutes
5. Satellites
6. 100 thousand million
7. Canis Major Dwarf Galaxy
8. 10s of thousands of years
How many moles of I2 are in 8.23 moles of NaI ?
Answers
Answer: 4.12
Explanation:
we know that the given mol is 8.23 mol and they are 2NaI and I2 so we will write the equation like this.
8.23mol NaI x 1mol of I2 ÷ 2molNaI = 4.115≅ 4.12 mol of I2
we placed NaI at the bottom to cancel out with the 8.23 mol of NaI
HELP PLEASE!! IF YOU DO AND ANSWER CORRECTLY YOU WILL GET A BRAINLIEST 5 STARSSSS AND A THANKS!! PLEASE PLEASE HELPPP
When scientists communicate about their work, what information should they make sure is included:
1.
2.
3.
4.
5.
6.
7.
Answers
2.) present at conferences
3.) present at universities
4.) inform popular media’s
5.) show data
6.) show hypothesis
7.) and give a theory
A sample of oxide iron weighing 2.40g was heated in a stream of hydrogen until it was completely converted to the metal. If the metal weighed 1.68g. What is the formula of the iron oxide? (Fe=56, O=16)
Answers
Answer:
Fe₂O₃
Explanation:
To solve this question we must find the moles of Iron in 1.68g. With the difference of the masses we can find the moles of oxygen. The formula will be obtained with the ratio of both amount of moles:
Moles Fe:
1.68g * (1mol / 56g) =0.03moles
Moles O:
2.40g-1.68g = 0.72g * (1mol/16g) = 0.045moles
The ratio O/Fe is:
0.045moles / 0.03moles = 1.5 moles. this ratio is obtained if the formula is:
Fe₂O₃Can anybody do this?
Answers
Answer:this should be c
Explanation:
Your car burns gasoline as you drive from home to school
Answers
I have a chemistry writing promp but it’s asking questions here the prompt: What is the empirical formula if you have 36.84% nitrogen and 63.16% oxygen? State three potential molecular formulas from the empirical formula in A. If the molecular formula mass in A is 760 grams/mol, what is the molecular formula? WIll the molecular and empirical formulas in A and D have the same characteristics? Why or why not?
Answers
Explanation:
1. Find the empirical formula of a compound that has 36.84% of Nitrogen (N), 63.16% of Oxygen (O).
Secondly, Find molar mass of each component: N=14.0067, O=15.9994
Thirdly, convert to moles: N=2.6301698472874, O=3.9476480368014
Then find smallest mole value: 2.6301698472874
Divide all components my smallest value: N=1, O=1.5009099282591
Then divide by fractional component of each mole value: N=1.9963668986866, O=2.9963668986866
Round to closest whole numbers: N=2, O=3
Lastly, combine to get the empirical formula:N2O3
Shontal compared some of the properties of a marble to a piece
of wood. She placed a marble and a piece of wood in a bucket filled
with water. Shontal observed the wood floating on top of the water,
but the marble sank to the bottom of the bucket. Which statement
best explains why the marble and piece of wood acted differently in
the water?
Answers
Answer:
I can't see your answer choices, but it would be something that related to density of the marble vs. wood.
Explanation:
Density is mass divided by volume. A marble with a greater density than water will sink, and wood with lower density than water will float.
A 1500 N force gives an object an acceleration of 3 m/s2.
What is the mass of the object?
Answers
Answer:
500kg.
Explanation:
Newton's Second Law relates force, mass and acceleration to one another through the equation:
F = ma
We can rearrange this equation algebraically to solve for mass:
m = F/a
Substituting our known values for the variables, we get:
m = 1500N/3m/s2
So mass of the object is 500kg.
We know it's kg because Newtons (N) are a unit defined as 1 kg/m/s2, so m/s2 cancels out when we divide :)
When you push or pull something, you are creating
Answers
force but could be tension
How many grams of NaN3 are required to produce 283 L of nitrogen gas about the size of an automotive air bag, if the gas has a density of 1.25 g/L? Please answer ASAP
Answers
3.300
bc im right fgdfgdffffffffg
a. An undesirable constituent of air
Answers
Complete Question:
Give one word for the following.
a. An undesirable constituent of air
b. The constituent of air that helps in burning
c. The thick layer of air surrounding the earth
d. Carbon dioxide does not support this process
Answer:
a. Pollutant.
b. Oxygen.
c. Atmosphere.
d. Combustion.
Explanation:
a. Pollutant: an undesirable constituent of air. Pollution can be defined as the physical degradation or contamination of the environment through an emission of harmful, poisonous and toxic chemical substances known as pollutants. Some examples of pollutant are carbon monoxide, nitrogen dioxide, ozone, etc.
b. Oxygen: the constituent of air that helps in burning. Oxygen is an element that aids in the combustion (burning) process because it is an oxidizing agent.
c. Atmosphere: the thick layer of air surrounding the earth. It comprises of nitrogen, oxygen, carbon dioxide, argon and other gases in very small proportion.
d. Combustion (burning): Carbon dioxide does not support this process but it is an end product of the process.
Combustion can be defined as an exothermic chemical reaction between physical substances, usually in the presence of oxygen and hydrocarbons to produce heat, light and carbon.
a 2.7 L of N2 is collected at 121kpa and 288 K . if the pressure increases to 202 kpa and the temperature rises to 303 K , what volume will the gas occupy?
Answers
Answer:
The gas will occupy a volume of 1.702 liters.
Explanation:
Let suppose that the gas behaves ideally. The equation of state for ideal gas is:
[tex]P\cdot V = n\cdot R_{u}\cdot T[/tex] (1)
Where:
[tex]P[/tex] - Pressure, measured in kilopascals.
[tex]V[/tex] - Volume, measured in liters.
[tex]n[/tex] - Molar quantity, measured in moles.
[tex]T[/tex] - Temperature, measured in Kelvin.
[tex]R_{u}[/tex] - Ideal gas constant, measured in kilopascal-liters per mole-Kelvin.
We can simplify the equation by constructing the following relationship:
[tex]\frac{P_{1}\cdot V_{1}}{T_{1}} = \frac{P_{2}\cdot V_{2}}{T_{2}}[/tex] (2)
Where:
[tex]P_{1}[/tex], [tex]P_{2}[/tex] - Initial and final pressure, measured in kilopascals.
[tex]V_{1}[/tex], [tex]V_{2}[/tex] - Initial and final volume, measured in liters.
[tex]T_{1}[/tex], [tex]T_{2}[/tex] - Initial and final temperature, measured in Kelvin.
If we know that [tex]P_{1} = 121\,kPa[/tex], [tex]P_{2} = 202\,kPa[/tex], [tex]V_{1} = 2.7\,L[/tex], [tex]T_{1} = 288\,K[/tex] and [tex]T_{2} = 303\,K[/tex], the final volume of the gas is:
[tex]V_{2} = \left(\frac{T_{2}}{T_{1}} \right)\cdot \left(\frac{P_{1}}{P_{2}} \right)\cdot V_{1}[/tex]
[tex]V_{2} = 1.702\,L[/tex]
The gas will occupy a volume of 1.702 liters.
HELP ME PLEASEEEEE!!!!!!!!!!
Answers
Answer:
I dont know what the answer is
Explanation:
An atom will be neutral if which of the following are true of its arrangement of subatomic particles?
The number of its protons is equal to the number of its electrons
All of its electrons orbit its nucleus at exactly the same distance
The sum of the atom's protons and neutrons is greater than zero
The nucleus of the atom contains more neutrons than anything else